Shock wave therapy was originally developed by Dornier MedTech to break up kidney stones nearly a quarter of a century ago. Since then, physicians discovered that Extracorporeal Shock Wave Therapy (ESWT) could benefit people with soft tissue and bone injuries.
The Dornier EPOS Ultra was FDA Class 3 approved for ESWT and the treatment of chronic plantar fasciitis. Other similar tendinopathy treatments now safely and effectively treated by ESWT are Achilles tendonitis, tennis elbow (lateral epicondylitis), shoulder tendonitis, patellar tendonitis, and plantar fascia arch pain (among others).
High Energy ESWT Dornier Epos Ultra is the only FDA device proven to repair damaged tendon or fascia. Recently we have seen some manufacturers of devices that are unfocused pressure waves trying to position their product as an ESWT machine. Pressure wave devices are used for relaxing muscles and trigger point pain. They are not ESWT, and do not product the same results or function in the body. Insist on High Energy ESWT with the Dornier Epos Ultra for a curative procedure.
What makes Excellence Shock Wave Therapy stand out is our commitment to our physicians and our patients. We are obsessed with providing a safe, comfortable and convenient treatment. We do everything in our power to follow through with you to cure your pain. Excellence is not simply our name, it is our promise.
Your condition is unique. We are here to help you find out if ESWT is right for you.
Contact us for a no pressure review of your case, an information packet, and an informational video.
I can't believe the difference! I had the operation on my other foot and went through a long recovery. This was terrific! I was back to normal very quickly.
”- Rita C., Paulsboro, NJ
ESWT is one of the few medical technologies that performs best on people with chronic conditions that did not respond to conservative traditional therapy. ESWT accelerates the body’s own healing process in a number of ways:
Your condition is unique. We are here to help you find out if ESWT is right for you.
Contact us for a no pressure review of your case, an information packet, and an informational video.
After Shock Wave Therapy in February, I took my time in healing. In late April I began to run again. I competed it my first 5K in June with a time of 30 minutes and my first Triathlon in July. Clearly I was able to resume all activities. I am a believer.
”- Nicole G., Robbinsville, NJ
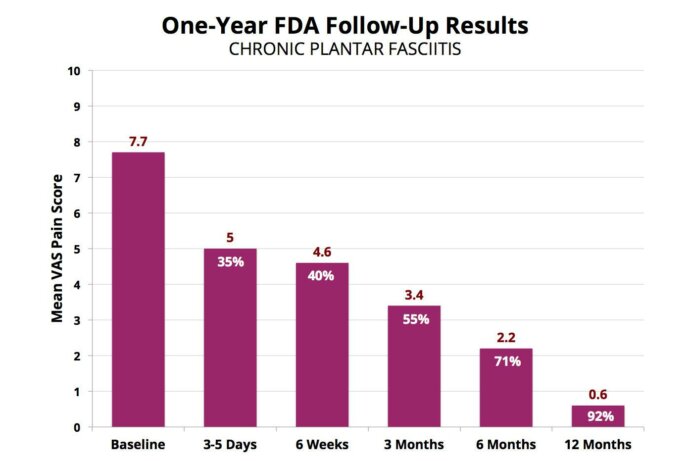
Our own experience and the FDA ESWT study using our treatment protocol shows an overwhelming success rate. In the study, a group of 150 patients was asked to rate their pain level on a scale of 1 to 10. The average pain level before treatment was 7.7.
After receiving one ESWT treatment, the patients were asked one year later to rate their pain level again on a scale of 1 to 10. The patients who reported had an average pain score post-ESWT of 0.6. This is a 92% reduction in pain and a much higher rate of success than any non-surgical or surgical procedure. More importantly, ESWT is very safe.